Scours Prevention Products
First Defense scours prevention products give you the greatest certainty of successful passive transfer, setting your calves up for a productive life. First Defense is easy to use. One dose at birth provides an instant dose of immunity from E. coli, coronavirus and rotavirus.

Tri-Shield® First Defense® Calf Scours Prevention
A single-dose gel tube and the only veterinary biologic with a guaranteed dose of immunity against E. coli, coronavirus, and rotavirus.
- A single-dose gel tube of Tri-Shield administered at birth, along with maternal colostrum, has been proven effective in third-party research to provide passive immunity against K99+ E. coli, bovine coronavirus, and rotavirus infections.
- Proven to provide passive immunity against the three most common scours-causing pathogens and reduce rotavirus shedding.
Approved by the USDA Veterinary Center for Biologics, Tri-Shield immediately delivers a guaranteed dose of passive antibodies to newborn calves. Unlike a dam- or calf-level vaccine that works to create an antibody response, Tri-Shield antibody therapy provides direct immunity.
“You can just compare calves that you have on First Defense compared to calves that aren’t. There’s definitely a big difference. Maybe not day one, but as they’re in their first three weeks when they’re in the calf hutches and they’re on milk. Once they start grain, we noticed a big difference on their milk and grain intakes, as well as just being healthier all around.”
– Zach Damrow, Seagull Bay Dairy, American Falls, ID

Reduces Rotavirus Shedding
A recent study by Skidmore et al* demonstrated that Tri-Shield significantly reduced rotavirus shedding in calves during the height of the disease, including those that achieved successful passive transfer from a quality colostrum feeding. As a result, you can limit the rotavirus pathogen in the calf environment, reduce the level of disease challenges herd mates are exposed to, and reduce the frequency and severity of scours outbreaks.
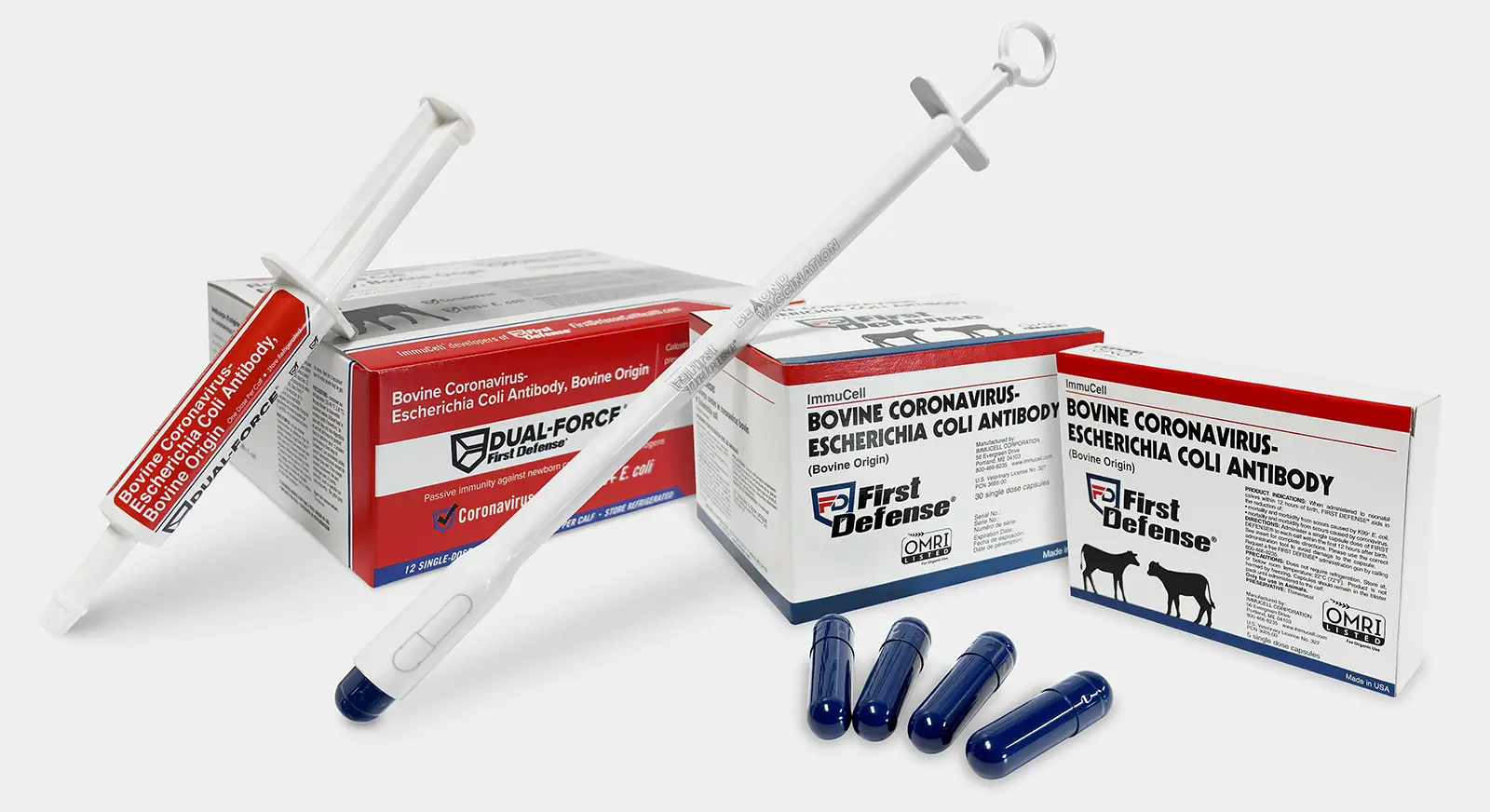
First Defense® Bolus and Dual-Force® Gel Tube for Calf Scours Prevention
An immediate guaranteed dose of immunity against E. coli and coronavirus infections, in a bolus or single-dose gel tube.
- Instantly deliver the passive antibodies newborn calves need to prevent scours.
- Jump straight to antibody protection while removing reliance on the common variable vaccine response of dam- and calf-level vaccines.
- One dose, a single bolus or gel tube, administered at birth gives each calf a guaranteed level of protection from E. coli and coronavirus infections.
Easy to Administer
The First Defense bolus can be easily administered with a balling gun. Our Dual-Force gel contains a non-digestible blue dye that serves as a marker, turning your calf’s first stool after administration a neon blue-green to help confirm proper administration.
“We have been using First Defense for the last 3 to 4 years and are really happy with the product. For us it’s important, it’s a big investment and a huge return. The product is convenient and versatile. Knowing we have protection as soon as the calf hits the ground is satisfying and comforting. This product gives assurance.”
– Dan Kullot, DVM, Syracuse Dairy, Syracuse, KS


First Defense Technology® Calf Scours Prevention
New and improved 90-dose bulk powder.
New First Defense Technology is based on the same technology as First Defense® boluses and now contains added colostral fat and bioactive proteins. It is concentrated to deliver a rapid source of targeted globulin proteins to newborn calves.
Administering First Defense Technology
- Add one level scoop of powder to your newborn calf’s first liquid feeding.
- Mix it with fresh, thawed colostrum; milk; water; or an electrolyte solution.
- Stir or shake until fully dissolved.
For optimal results, administer within 24 hours of birth or during periods of stress.
“Prior to adding First Defense to our regimen, our calves would always run into problems with salmonella after having scours seven to 10 days after birth. We have been using First Defense at Cal Poly State University’s dairy unit and have seen calves getting through the first two weeks of life stage really successfully. We would recommend First Defense to anyone. This product is simple to administer and produces results.”
– Rich Silacci, Cal Poly State University, San Luis Obispo, CA

Approved by the USDA Veterinary Center for Biologics
First Defense products are approved by the USDA Veterinary Center for Biologics, the same agency that approves scours vaccines.
To learn more about First Defense scours prevention products, talk to your representative.
